|
Learned in other videos that the atom is in fact made up of even smaller constituent particles which is pretty amazing because atoms are already So in the sun it isn't really a case of helium combining with hydrogen in a reaction sense, rather they are just in close proximity to each other in a mixture. The main nuclear reaction occurring in the sun are series of nuclear reactions called the proton-proton chain (because it begins with the collision of two hydrogen atoms containing a single proton each) which eventually results in helium atoms.īut once helium is formed, it doesn't really combine with hydrogen afterwards in either chemical or nuclear reactions. Essentially what happens in the sun is that atoms of hydrogen-1 (that is isotopes of hydrogen with a single proton only) collide into each other with such force that they fuse into new atoms. Nuclear reactions are different from chemical reactions in that chemical reactions only involve the electrons of atoms and do not touch nuclei of atoms where we find protons and neutrons while nuclear reactions involve the nuclei of atoms. In the sun we don't really have chemical reactions involving helium, rather nuclear reactions. Helium doesn't really react chemically because its valence shell is filled and it is stable in its natural electron configuration. Reactions where the valence electrons of atoms interact to form new chemicals by breaking and forming chemical bonds. Usually in chemistry we're concerned with chemical reactions. The 2p orbital is closer to the nucleus than the 3s orbital, because it is in the second shell, which is closer to the nucleus than the third shell. 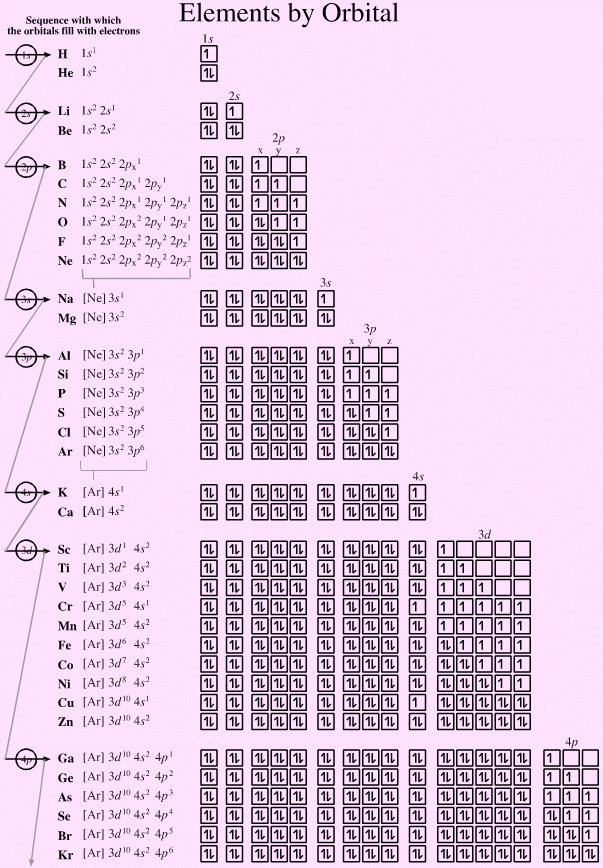 
In the third shell we again find p and s orbitals. Them comes the third shell even further away from the nucleus. But the 2s is of course further away from the nucleus, because it is in the second shell. In the second shell there are s and p orbitals. So in the first shell there is only one subshell, the s orbital. Now let's have a look at each shell in detail. The smallest, nearest to the nucleus is shell number 1. Imagine shells around the nucleus, that get bigger and bigger. This is also due to the history when they were discovered. We classified the different Orbital into shells and sub shells to distinguish them more easily. Similarly, the atoms of all group 7 elements have similar chemical properties and reactions to each other because all of them have seven electrons in their outer shell.An orbital is a space where a specific pair of electrons can be found. The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer shell. The electronic configurations of the elements in group 1: So, elements in the same group have similar chemical properties because they have the same number of electrons in their outer shell. When atoms collide and react, it is the outer electrons that meet and interact. The electronic configurations of atoms help explain the properties of elements and the structure of the periodic table.  
has an atomic number of (2 + 8 + 1) = 11Įlectronic configurations and properties of elements.The electronic configuration of sodium (2.8.1) shows that sodium, Na: the number of electrons in all shells of an element is represented in the periodic table as the element's atomic number.the number of electrons in the outermost shell of an element is represented in the periodic table as the group number that element is situated in.the number of circles in the electronic configuration of an element is represented in the periodic table as the period number that element is situated in.The electronic configuration of an element is related to its position on the periodic table. Electronic configurations and the periodic table
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
